So What Is Alkalinity?
Alkalinity is basically the resistance of a solution to acid. In the case of swimming pools, the pool water is the “solution” we are talking about.
Having proper alkalinity levels will help to manage and settle the Ph of your pool, makes chlorine more effective, and keeps your pool from becoming too corrosive or forming scale.
Its primary function is to act as a buffer for your pools alkalinity, preventing swings in Ph levels that would otherwise require frequent adjustment.
These swings are called the “Ph bounce” and it becomes a nightmare to manage, especially in salt chlorine pools as they have a tendency to naturally increase a pools Ph.
The buffering effect acts as a soft pillow absorbing those bounces and stabilizing off a pools Ph level making management much more predictable and less frequent in most pools.
What Is the ideal range for alkalinity in a pool?
The ideal level for a pools alkalinity is between 100-140 ppm.
This range ensures an appropriate buffer and some positive effects for chlorine. Calcium hypo-chlorite and sodium hypo-chlorite are expected to perform a little better in the lower end of this range with di-chlor, tri-chlor and chlorine gas to perform better towards the top end of the spectrum.
For the typical home pool, there’s no real benefit to micromanaging this far though so we simply recommend you stay within the 100-140 ppm range.
Any reading at/below 60 ppm, or above 180 ppm for almost all pools will require immediate adjustment. These levels are likely to greatly affect a pool’s LSI reading putting your surface and equipment at risk of failure.
Standard alkalinity test will give you your total alkalinity, which is almost what you need. In addition to knowing your total alkalinity, you should also know your carbonate alkalinity level.
Subtracting carbonate alkalinity from your total alkalinity is necessary to find your corrected or true total alkalinity which is the level you want to adjust.
There are two ways to find this carbonate alkalinity reading.
Method 1:
Total Alkalinity ppm – (Cyunaric acid ppm / 3) = Carbonate alkalinity
*this method assumes you alkalinity is between 7.2-7.8 and isn’t exact.*
Method 2:
Total Alkalinity ppm – (Cyunaric Acid ppm x [correction factor of pH]) = Carbonate Alkalinity

Once you know your Carbonate alkalinity, subtract this from your pools total alkalinity you got from your initial testing and you now have your pools adjusted total alkalinity.
This is the reading you’ll want to adjust to within 100-140 ppm total.
How To Adjust Your Pools Alkalinity
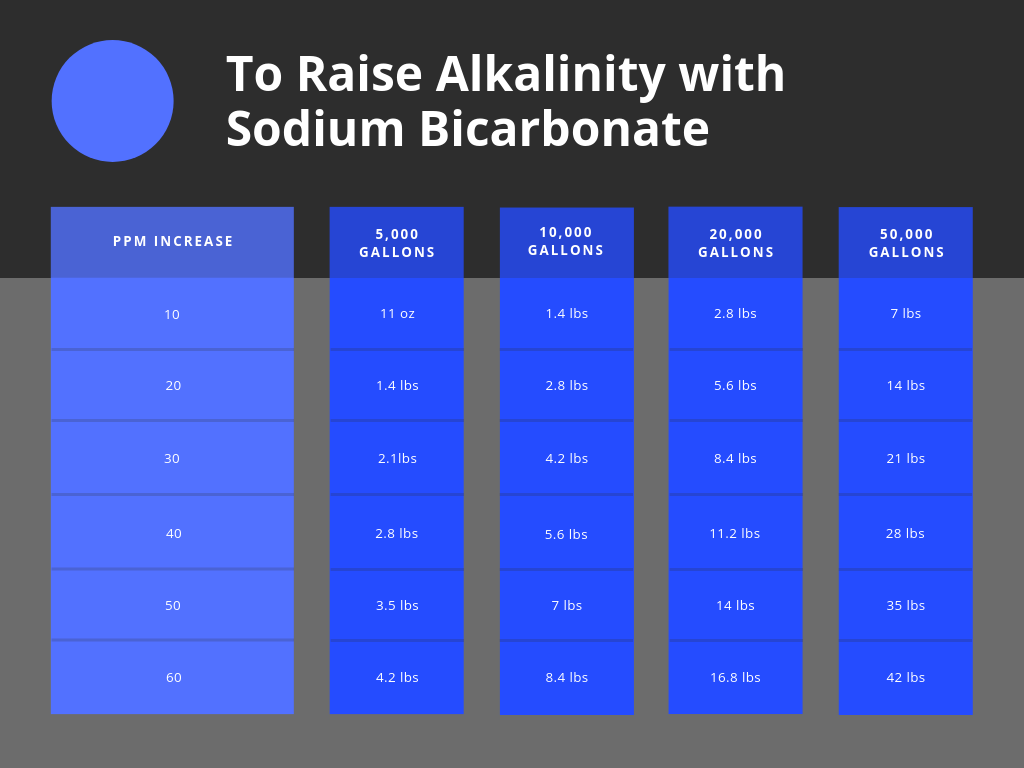
How To Raise Alkalinity In A Pool
Using Sodium bicarb is really simple. just measure out the weight you need and broadcast it over the water surface. Any chunks of bicarb that fall to the floor of the pool are simple enough to just brush to help break up and aid in the dilution, but it’s generally harmless to the surface of a pool.
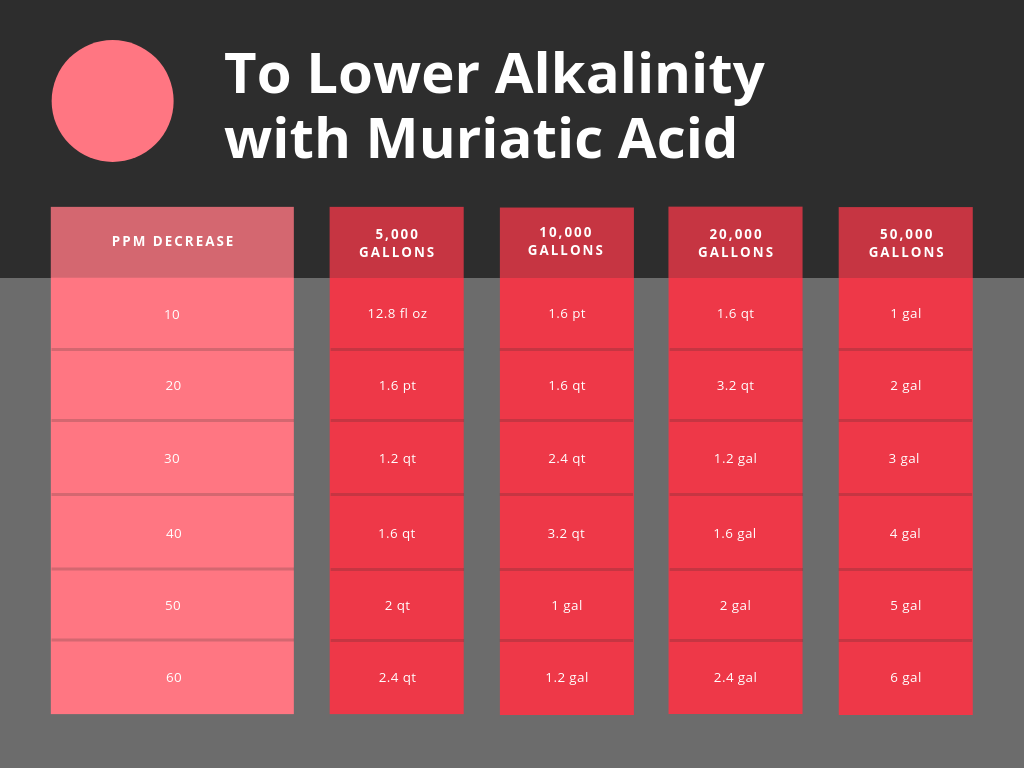
How To Lower Alkalinity In A Pool
There are multiple chemicals used to lower alkalinity, but the most common is muriatic acid.
To administer muriatic acid we recommend pouring this around the perimeter of the pool and avoiding “columning” the acid. We discussed muriatic acid safety in our Ph article here.
If you would like to dilute muriatic acid prior to pouring into the pool, we recommend diluting in an already filled bucket of water in a 4:1 ratio. this can make it safer to handle in large quantities.
*Any excessive changes of the alkalinity of a pool will have a knock-on effect to its Ph level. Read here if you need to know how to adjust the Ph of your pool.
Alkalinity Knock-on Effects
Minimal adjustments in alkalinity generally have minimal effects of Ph but large changes can drastically affect Ph readings in the days after an adjustment.
This is usually more the case for lowering alkalinity as the volumes of acid required are very high, causing a drastic shift in Ph. Allow a couple of days or until Ph stabilizes before attempting top readjust to the correct range.
F.A.Q.
Use muriatic acid. 1.6 pints will reduce 10,000 gallons of waters alkalinity level by 10 parts per million.
Use Sodium Bi-carbonate or baking soda. 1.4 lbs will raise 10,000 gallons of waters alkalinity level by 10 ppm
It’s is used to resist changes in Ph and is a vital part of proper pool chemistry balance
between 100-140 ppm is ideal. Lower than 60ppm or above 180ppm Requires Immediate Adjustment








